medical outcomes
QL-Recorder?

Examples of successful applications
This page informs you briefly about work done recently using the Quality-of-Life-Recorder.
Please note: This page has not been updated after 2010.
2010
Deployment of the QL-Recorder in a multinational research project
- University of Ulm, Günzburg, Prof. Dr. Reinhold Kilian, Dr. Prisca Weiser - Generation of a multilingual set of multiple electronic questionnaires for the HELPS project.
Deployment of the QL-Recorder in institutional routine patient care
- Reha Zentrum Bern, Heiligenschwendi, Switzerland, PD Dr. med. Armin Stucki.
- Clinic for child psychiatry and psychotherapy, University of Marburg, Germany, Prof. Dr. Fritz Mattejat, Prof. Dr. Katja Becker, Dipl. human. biol. Udo König.
- Radiation clinic, University of Erlangen, Germany, Dr. Ulrike Keller.
Lilly Quality-of-Life Prize 2009
Conception and continued development & support of the Quality-of-Life-Recorder have been awarded the Lilly Quality-of-Life Prize 2009.
Thank you for this kind recognition and support of my work!
2006 and 2007
Comparison of the SF-36 in Chinese Language in paper-and-pencil vs. QL-Recorder based versions
Chen T, Li L, Sigle J, Du Y, Wang H, Lei J: Crossover randomized controlled trial of the electronic version of the Chinese SF-36.
J Zhejiang Univ Sci B 2007 8(8):604-608. ISSN 1673-1581 (Print), ISSN 1862-783 (Online).
www.zju.edu.cn/jzus
www.ncbi.nlm.nih.gov/pubmed/17657865 (Publication available online, free full-text in PubMedCentral)
www.springerlink.com/content/3374746645382p87
A paper-and-pencil version and a version based upon the QL-Recorder with AnyQuest for Windows were administered to 100 students and 50 patients. The electronic version was very well accepted and collected data were similar.
Project (No. WKJ2006-2-016) supported by the project of "Effect of Chronic Disease and Health-Related Quality of Life on Health Service Utilization" from the Ministry of Health, China.
Usage of the QLQ-C30 and the SGRQ on the QL-Recorder linked to practice management software via WLAN in general practices (family practices)
Rogausch A, Sigle JM, Seibert AJ, Thüring S, Kochen MM, Himmel W:
Feasibility and acceptance of electronic quality of life assessment in general practice: an implementation study.
Health and Quality of Life Outcomes. 2009 7:51 doi:10.1186/1477-7525-7-51. PMID: 19493355
www.hqlo.com/content/7/1/51 (Publikation online verfügbar, Volltext frei zugänglich).
Rogausch A, Sigle J, Thüring S, Kochen M, Himmel W: Patientenbefragung per Tablet-PC.
Dtsch Arztebl 2007; 104(42): A 2857-60
www.aerzteblatt.de/lit4207 (Publication available online, full-text freely accessible).
A project of the Department for General Practice at the University of Goettingen studies technical and organisational possibilities regarding the administration of electronic quality of life questionnaires in patient's waiting time (additional information below - 2005.)
Supported by the Ministery for Education and Research (BMBF) within the project "Research in general practice", grant no. 01 GK 0201.
Validation of an electronic version of the SIRO (eSIRO) for screening before psycho-onkologic treatment during radiotherapy
Herschbach, Berg, Pirker, Henrich, Emrich, Sehlen, Dühmke, Geinitz, Molls: Computer based evaluation of a screening procedure for psycho-oncological treatment during radiotherapy
In: Abstracts of the 8th World Congress of Psycho-Oncology, 16 th-21 st October 2006, Ferrara-Venice, Italy
Psycho-oncology 2006; 15(iss 2 Suppl): S1-462; PMID: 17338072 (Overall)
www.ipos2006.it
www.krebshilfe.de/archiv...
www.med.tu-muenchen.de/...
The paper version of the SIRO was administered 131 times, the electronic eSIRO was administered 142 times. 99% of the patients find the electronic version very easy to use - versus 96% for the paper version (not significant). The eSIRO results are computed immediately and can be integrated in the workflow. Time and cost requirements favour the electronic version; and staff confirm that the paper-and-pencil version requires more resources. Questionnaire results are similar for both versions.
In addition to the usual patient interface, a fast scoring tool for paper questionnaires was integrated for this project. The complete eSIRO is available on CD-ROM. The complete documentation is available in "Docs+Support" on this site (in German).
Supported by the Deutsche Krebshilfe e.V., grant no. 106079.
Comparison of a paper version and a QL-Recorder based electronic version of the Swedish QLQ-C30 at the University of Uppsala
Once again, usage of the QL-Recorder at the Gynecologic Clinic of the University of Graz, to assess the psychosocial situation of pregnant women
Selected application examples until 2005
BMBF funding for a QL-Recorder application
A study was started at the Department of Family Practice of the University of Goettingen, in which the QL-Recorder is used in 25 general practices, funded by the BMBF (German Ministery of Education and Research). Within this study, the interface of the QL-Recorder within German practice management systems via the xDT interface via WLAN (Wireless Local Area Network, GDT tools available on this site) will be adopted to a broader variety of vendors.
Clinical study in patients with COPD at Rolle, Switzerland
Managed by Dr. Oliver Staneczek, the QL-Recorder was used to administer several questionnaires in a clinical study in a group of severely ill patients with COPD at the hospital in Rolle, Switzerland.
Use at the University of Graz, Austria
Managed by Prof. Dr. Eva Greimel, the QL-Recorder was used at the Gynecologic Clinic of the University of Graz to evaluate several questionnaires (King's Questionnaire, SF-36, HADS).
Use at the Western General Hospital, Edinburgh, Scotland
Managed by Robert Rush and colleagues, the QL-Recorder was used at the Western General Hospital in Edinburgh for routine administrations of patient questionnaires, e.g. with regard to screening for psychologic risk factors in oncology patients. Published results:
December 2002
The Lilly Quality-of-Life Research Award 2002 - a QL-Recorder users's experience!
A project started by Dr. Lutz Goldbeck at the University of Ulm two years ago, which uses the QL-Recorder as central data collection tool in patients with Cystic Fibrosis, has received the Lilly QoL Research Award 2002. Further information is available on the Multimedia documents page.
November 2002
Presentation of the QL-Recorder at the MEDICA, Düsseldorf, by the Fraunhofer Gesellschaft IAO
Based upon their own positive experience, the Institut Arbeitswirtschaft und Organisation of the renowned Fraunhofer Gesellschaft chose the QL-Recorder as an example of innovative technology useful to face the challenges emerging from contemporary changes in the field of medicine. Further information is available on the Multimedia documents page.
August 2001
Presentation of the QL-Recorder at the University of Geneva
A Palmax PD-1000 based QL-Recorder was presented to several colleagues at the University of Geneva and found a very positive echo.
In the hospital of Rolle, four pneumologic patients were presented a variety of questionnaires (either: demographics, MRF-28 and SGRQ, SF-36, HADS, or: demographics, EORTC QLQ-C30, MRF-28, HADS; all in the French language) to gather practical experiences. For this purpose, the QL-Recorder was brought into each patient's room. After the assessment, it was collected, brought back to the medical assistants' room, where the printouts were generated on a printer using the infrared port.
The participating patients were two ladies and two gentlemen, in the sequence f, m, f, m, aged 69, 72, 78 and 82 years. In the first two sessions, we used among other questionnaires, the SF-36. The results for PCS were 21 and 23, for MCS 53 and 58 (reference: US general population). In the last two sessions, we used among other questionnaires, the EORTC QLQ-C30. The results for Physical Function were 87 and 40; for Cognitive Function 83 and 0; for Dyspnea 67 and 100. HADS results were between 2 and 11; other results can be provided upon request.
During the first two measurements, at least one of us stayed in the patient's room to look for and record any difficulties or to help if required. During the third measurement, we left the patient alone for some time, and during the last measurement, we left immediately after we saw he had answered the first two questions. The durations of questionnaire administration were (hr:min:sec) 1:04:10, 0:32:27, 0:16:36 and 0:29:23 respectively; the first patient thought very carefully about each question.
She also read aloud some of the questions across the room with her room-mate and stated that it would be difficult to decide between just "yes" or "no", while the room-mate of another patient looked across his shoulder for some time. In real life research, patients should of course be carefully advised to avoid both situations, and neither should a staff member or relative be too close.
The patients found the electronic questionnaire interesting and were happy to participate. They had no difficulties at all with the device (but very few with the content of some questions from the specific instruments; details available upon request) and all of them responded, they would prefer this kind of questionnaire presentation versus a paper form. Among other advantages they found that there were no difficulties with getting lost in multiple lines often printed on forms, and said it would be just more fun to fill in an electronic questionnaire instead of a paper form. To provide information to their doctor, they found a questionnaire advantageus, because first, less would be forgotten, and second, they could take their time to reflect each item just as required, while during the consultation, they would not want to keep their doctor for too long. The fact that during the questionnaire administration, no staff was attending, did not disturb them at all but was perceived as positive instead for the same reason. We asked every patient how the technical solution could be improved. However, we received no suggestion at all; instead we were affirmed that (verbatim, and multiple times) the current version was perceived as absolutely perfect. There were some comments regarding the contents of the questionnaire. A combination of QLQ-C30, a short indication specific instrument and HADS seemd to be most comfortable for patients and at the same time very informative for the clinician. The possibility to display some questions (for example regarding the impact of having to use respiratoritary assisting devices) only conditionally (after having asked whether such a device has been used) has proven to be very useful. We discussed the generated printouts with some of the patients, this was also received very positively.
We also tried out a small wireless network (WLAN) using the PD-1000 and a laptop equipped with a second wireless LAN adaptor (Aironet), but we found that the link could not reach from the assistants' room to the last rooms of the corridor. We did not spend more time on trying with differently positioned devices.
Personally, I enjoyed this time at the lac Léman a lot - thanks to Dr. Olivier Staneczek for his kind invitation!
May 2001
Integration of the QL-Recorder in a general practitioners IT environment
Using the newly prepared GDT interface of the QL-Recorder, it was seamlessly integrated into a general practitioners IT infrastructure. The existing infrastructure uses QuincyPCnet by Frey ADV GmbH to provide an electronic medical record. The general practitioner (and myself) can now request a routine Quality-of-Life assessment from the electronic medical record for the currently selected patient. Immediately, the QL-Recorder located in a separate room displays the patient's name and date of birth and asks for a confirmation to begin the assessment. The patient answers all questions by himself, using a nice touch-screen. Computed questionnaire results could be printed immediately and automatically, including the patient's course over time - but we do not print them, because they are transferred into the lab results page of the electronic medical record immediately, and QuincyPCnet presents them in the usual tabulated way alike any other lab result :-)
As Quality-of-Life measurement results are stored together with other medical data inside the electronic medical record, clinical and outcome or patient benefit related information may now be evaluated in combination without additionally incurring workload. This makes outcome oriented research in routine care and outcome oriented quality assurance measures finally feasible.
April 2001
Spring meeting of the EORTC Quality-of-Life Group:
A QL-Recorder workshop for group members achieved a very positive echo. An arrangement was agreed upon which includes the following key points:
- The eQLQ can be provided to users, who have registered with the secretary of the EORTC for academic use of the QLQ.
- For this purpose, the EORTC WWW site for registration will list the eQLQ in addition to paper based modules. Who requests the eQLQ will be provided with a link to a WWW site from where the eQLQ can be downloaded.
- The WWW based software library of the QL-Recorder will direct visitors interested in the eQLQ to the registration site of the QoL Group. The information CD will include a time-limited version of the eQLQ showing a clear remark requesting registration.
- The QL-Recorder information CD is distributed to all members of the group.
January 2001
EORTC Quality-of-Life Group: Pilot testing of customised packages
A customised configuration of AnyQuest for Windows, including an electronic version of the EORTC QLQ-C30 Version 3.0, was prepared for pilot testing by members of the EORTC QLG.
January 2001
The Fraunhofer-Institut für Arbeitswirtschaft und Organisation uses the the QL-Recorder to administer a patient questionnaire in a clinic; collected data are transferred through the Internet
A customised configuration of AnyQuest for Windows, including several questionnaires, was generated for the Fraunhofer-Institut für Arbeitswirtschaft und Organisation, Stuttgart, and installed on a Palmax PD-1000 mini-pen-computer. The system is used in a clinic to present questionnaires to patients. Collected data can be packaged and encrypted automatically, and sent through the Internet for central evaluation.
November 2000
Import-Interface for the tumor documentation system TREG updated, electronic Quality-of-Life questionnaire linked to tumor documentation system in Bremen
The import-interface of T-REG was updated, now it can also import data collected according to the EORTC QLQ-C30 Version 3.0 with AnyQuest for Windows. In the Zentralkrankenhaus Ost, Bremen, a Palmax PD-1100 used as QL-Recorder was connected to the network, so that Quality-of-Life-data collected directly from patients can be imported into the tumor documentation system without any manual steps in between. In the database of the tumor documentation system, they are available for interactive statistical analysis immediately.
October 2000
Glaxo Wellcome plc.: eIBSQOL Version 2.0
For the UEGW 2000 in Brussels, the eIBSQOL was updated and additional languages were added on behalf of Glaxo Wellcome plc. - Currently, it is available in 19 language versions, including Greek.
November 1999 - July 2000
GZW: Data collection for validation of questionnaire: QoL in geriatric patients with dementia
In the Geriatriezentrum am Wienerwald, Lainz/Vienna, a data collection was carried out for the validation of a newly developed questionnaire regarding quality-of-life of old-old patients with dementia. Different configurations for nurses and physicians were prepared on two Palmax mini-pen-computers, including 9 questionnaires. 771 measurements were collected. This international project is based upon a co-operation with colleagues from the Universities of Umea, Sweden, Graz, Austria, and Ulm, Germany.
October 1999 - Januar 2000
The QL-Recorder in an RCT
Complementing physical parameters, the QL-Recorder was used in a randomized controlled trial to assess QL as a therapy outcome. Details will follow after publication.
November 1999
Glaxo Wellcome plc.: eIBSQOL
At the UEGW 1999 in Rome, Glaxo Wellcome plc. have presented a new drug together with its corresponding outcome measurement tool: eIBSQOL uses QL-Recorder technology to make an electronic version of the IBSQOL questionnaire available to physicians in 10 languages for immediate use in routine practice.
November 1999
More than 20.000 assessments made
A survey at some of the known users of the original and current versions of the LQ-Recorder show that certainly more than 20.000 times patients have filled in an electronic questionnaire with this instrument. A report on individual projects and results will be prepared during the next months.
May 1999 - May 2000
University of Ulm, Pediatric Clinic: Data collection for validation of questionnaire: QoL of Children
In the Pediatric Clinic of the University of Ulm, a data collection was carried out for the validation of a newly developed questionnaire regarding quality-of-life in children. Two configurations, including questionnaires for parents and children, respectively, were prepared on a Palmax PD-1000 for this project.
1999
Import interface for the hyperthermia therapy documentation system FEBROSKRIPT
Stefan Heckel Hyperthermiesysteme, Esslingen/Germany, developed an import interface for QL-data recorded with AnyQuest for Windows into their hyperthermia therapy monitoring and documentation system FEBROSKRIPT. Among other possibilities, the solution gives users the possibility to access all recorded data, including some medical documentation, physical parameters and QL-data for basic analysis.
1998/1999
Import interface for the tumor documentation system TREG
Dr. Friedrich von Bültzingslöwen had an import interface for QL-data recorded with the original QL-recorder into the tumor documentation system TREG developed. Among other possibiliterat/lities, the solution gives users the possibility to access all recorded data, including medical documentation and QL-data for immediate and interactive statistical analysis.
1998/1999
Import interface for the Psychotherapy-Quality-Assurance-System AKQUASI
In co-operation with the Forschungsstelle für Psychotherapie Stuttgart (FOST), import interfaces for questionnaire data from scanners and from the LQ-Recorder into AKQUASI were developed. For a number of questionnaires used with AKQUASI, questionnaire-definition-files for the QL-Recorder were generated. This solution can reduce the workload associated with the use of questionnaires for quality assurance significantly.
1998
Presentation of the QL-Recorder at the CeBIT
The QL-Recorder using AnyQuest for Windows was selected to be presented as one of three projects from the University of Ulm at the stand of the country of Baden-Württemberg.
1997-1999
Evaluation of courses during medical training and continued education with AnyQuest for Windows
Continued education courses of the AG Klinische Ökonomik in Evidence-Based Medicine and Clinical Economics were evaluated regularly using the QL-Recorder. Immediately after the last session, an objectivized feedback from participants in several dimensions (e.g. preparation of teachers, interest in the topic before and after the course, motivation and satisfaction of participants) was available.
We also evaluated courses within our teaching activities for medical students. Here is a report (German) containing a complete evaluation of a course given for a 9th/10th semester including thoughts about possibly required changes in our ways education system.
09.01.1997
Alan Coates: More experiences
Today, Alan used the system with 4 more patients as time allowed. He found that the Fujitsu Stylistic 1000 can operate on one battery charge during the whole work day, going into standby mode between patients. A french speaker used the system without difficulty. And - sadly, say I - my colourful rainbow in the background reduces readability for patients with impaired visual acuity, so it should rather be turned off if this is critical...
Please note: I do not know about any reference study comparing results from measurements with and without colourful questionnaire paper!
08.01.1997
Alan Coates, Dr. med. Jörg M. Sigle: A maiden voyage: First use of AnyQuest for Windows by patients & data transfer through the Internet
Prof. Alan Coates, medical oncologist from Sydney (review the literature references and AnyQuest's help file...), tried out the Quality-of-Life-Recorder to assess patients' quality of life for the first time during his out-patient clinic in Dubbo.
He used AnyQuest for Windows V. 2.15 beta on a Fujitsu Stylistic 1000 with a color display and Windows 95, and the questionnaire definition file for the EORTC QLQ-C30. He wrote me a report in an e-mail and sent the exported •.DIF-file with the results to me to have a look at it.
According to his report, because he had no assistant, 11 consecutive patients were offered the electronic questionnaire by himself after the end of each consultation. The questionnaire was generally well received, and some of the patients were very impressed by the system.
The patients were a very varied lot. One who uses computers every day, liked the system very much. Another, 75 year old with a brain tumour, had never used any computer, but had no difficulty. One very nervous woman had a shaky hand and repeatedly "double clicked" both when entering her name (which frustrated her considerably, though she used <<BACK to retrieve the errors) and more importantly in the boxes. Under this circumstance the program cancelled the check mark, but moved immediately to the next question, so that at the end she had 11 questions unanswered. She did better when instructed on how to hold the stylus.
After I had received the data, I imported them into Microsoft Excel to do some minimum statistics. Afterwards, I sent the complete •.XLS file back to Alan who could read it again without any difficulties.
I would like to present some of the data from the experiment here:
The patients were 39.7, 39.8, 46.4, 47.3, 56.7, 57.1, 57.4, 61.6, 68.0, 68.3 and 75.4 years old, respectively, with an average age of 56,1 years..
The time to complete the questionnaire ranged from 2:20 (min:sec). to 7:10 with an average time of 3:44.
The following diagram shows the average results in the dimensions of the EORTC QLQ-C30 (it has been kept simple to keep Internet load low):
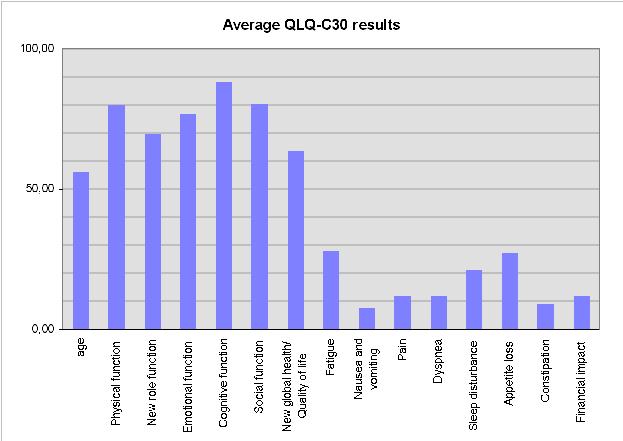
Of course, Alan could have done the data import and analysis on his own, but this way we demonstrated: downloading of the software from my WWW-Site, installation on the pen-computer, remote data collection with untrained patients using the electronic questionnaire, data transfer to centralised analysis and distribution of the results via Internet.
So we both think that this was a very successful maiden voyage...